Merck & Co. Inc, or Merck, has been a leading technology and science corporation making high-quality medicines and vaccines for over a century. The company is known for innovating and developing medicines and vaccines and delivering them worldwide through contracting. Read this to learn more about Merck & Co.’s contracts with the government.
Table of Contents
1. Merck & Co. wins U.S. Government COVID-19 Drug Molnupiravir Procurement, $1.2 Billion

The U.S. government contracted Merck & Co for testing and manufacturing molnupiravir. The MOVe-OUT Phase 3 clinical trial tests molnupiravir in non-hospitalized individuals with laboratory-confirmed COVID-19 and at least one risk factor for poor disease outcomes.
In this contract, Ridgeback Biotherapeutics and Merck will develop molnupiravir. Merck will receive roughly $1.2 billion to provide approximately 1.7 million courses of molnupiravir to the government once the Food and Drug Administration (FDA) grants an Emergency Use Authorization (EUA) or approval for molnupiravir. With risk capital invested in the development and increased production of molnupiravir, Merck anticipates having more than 10 million therapy courses accessible by the end of 2021.
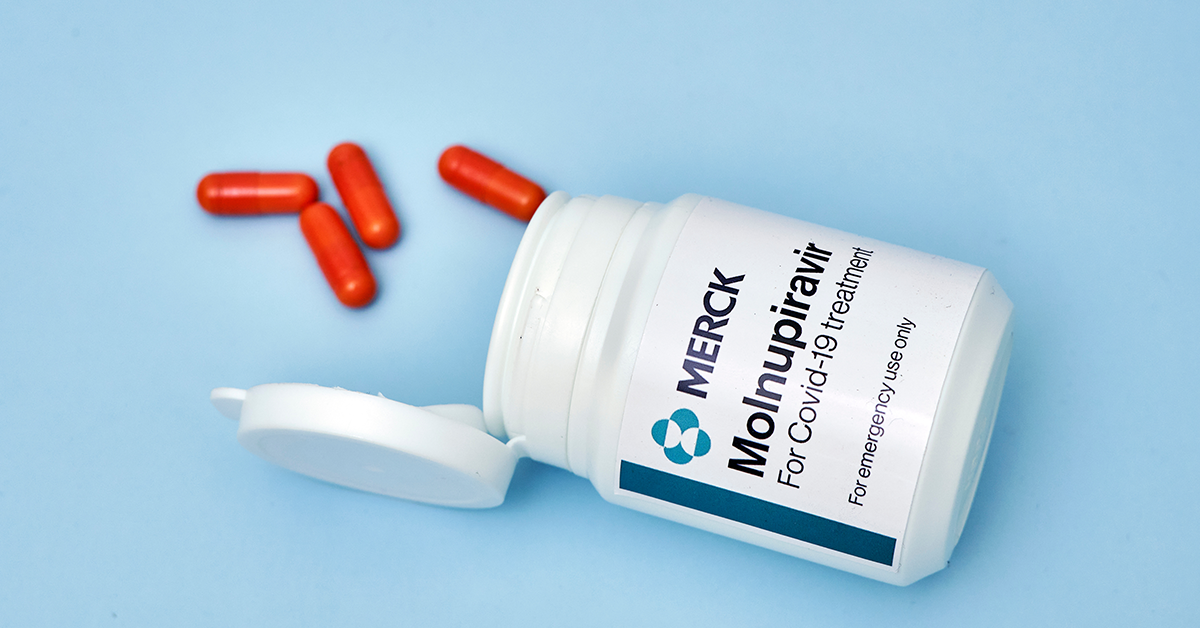
About Molnupiravir
Investigational oral antiviral candidate molnupiravir has been shown to inhibit the replication of SARS-CoV-2, the virus responsible for COVID-19. According to the Phase 3 MOVe-OUT clinical trial, the suggested dosage of molnupiravir, once authorized or approved, will be 800 mg twice daily for five days.
However, there are precautions with Molnupiravir. It is still not approved for Covid-19 patients 18 years old and below. The medication is also not advised to take as prevention for COVID-19 or take more than five consecutive days.
2. U.S. Government Additional 1.4 Million Courses of Molnupiravir, $2.2 Billion

The FDA granted an Emergency Use Authorization (EUA) or approved Molnupiravir; the United States government will exercise two options to buy an additional 1.4 million courses from Merck worth $1 billion in collaboration with Ridgeback Biotherapeutics.
The purchasing activity will happen between authorization and early 2022. The U.S. government can buy more than 2 million more courses through other options in the contract. Adults at high risk who have mild to moderate COVID-19 are the primary receiver of this developing medication.
3. Merck clinched a U.S. Government Contract to Build a New Lateral Flow Membrane Production Facility, $136.7

In Darmstadt, Germany, Merck’s Life Science business sector, MilliporeSigma, received a contract award worth €121 million or approximately $136.7 million last December 29, 2021. The U.S. Department of Defense, on behalf of the U.S. Department of Health and Human Services (HHS), requested to secure the local supply and production capacity of lateral flow membrane to ensure pandemic preparedness.
Since the epidemic began, the global demand for quick diagnostic manufacturers’ lateral flow membranes test has increased. The Nasal Swab Novel Coronavirus (2019-nCoV) Antigen Test or Lateral Flow Method is a quick test for identifying the N protein antigen of novel coronaviruses in a nasal swab sample.
Under the terms of the agreement, Merck will build a lateral flow membrane production plant, providing its customers with a greater supply of Hi-FlowTM Plus lateral flow membranes.
About Merck & Co. Inc

Merck & Co., Inc. is a research-focused biopharmaceutical corporation in the world. The company innovates new medical solutions to help hospitalized patients and offers widespread access to people to cure modern diseases.
Rob Davis is the current Merck & Co Inc chief executive officer and president. Rob also serves on the board of Project Hope, which empowers medical practitioners to provide better healthcare.
Merck is headquartered in Kenilworth, New Jersey, and has 110 locations in the United States.
Merck & Co. Inc FAQs
What do Merck & Co. do?
Merck develops novel pharmaceuticals, vaccines, biological therapies, and animal health products. The company aims to increase access to medicines and vaccines and improve the health and well-being of individuals and animals globally.
What other medicines does Merck make?
In the pharmaceutical industry, Merck has had several hugely successful new medicines. Some of the most notable drugs produced by the company are Clarinex, Fosamax, Implanon, and NuvaRing.
Is Merck & Co an American company?
Merck is an American pharmaceutical firm known worldwide for providing medical care services and products. The corporation is called Merck Sharp & Dohme (MSD) outside the United States.
Merck’s Continual Dedication to the COVID-19 Project
Since the beginning of the epidemic, Merck has been dedicated to researching SARS-CoV-2 infection and developing a comprehensive COVID-19 treatment. Merck’s leadership is continuing to produce excellent antiviral medicines through advanced research.







